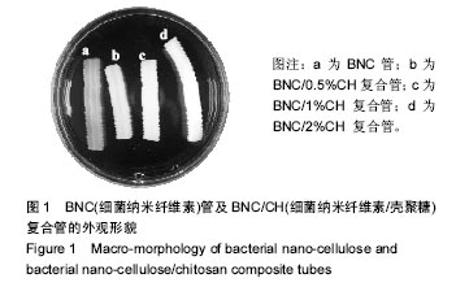
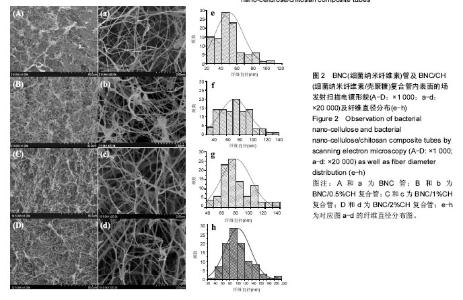
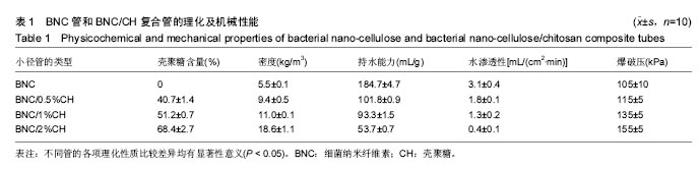
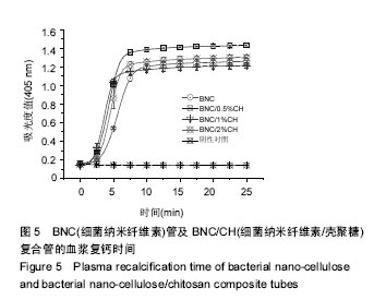
Chinese Journal of Tissue Engineering Research ›› 2017, Vol. 21 ›› Issue (34): 5467-5473.doi: 10.3969/j.issn.2095-4344.2017.34.010
Previous Articles Next Articles
Preparation and evaluation of bacterial nano-cellulose/chitosan composite tubes as potential small-diameter vascular grafts
- College of Chemistry, Chemical Engineering and Biotechnology, Donghua University, Shanghai 201620, China
-
Received:2017-07-24Online:2017-12-08Published:2018-01-04 -
Contact:Hong Feng, Professor, Doctoral supervisor, College of Chemistry, Chemical Engineering and Biotechnology, Donghua University, Shanghai 201620, China -
About author:Li Xue, Master, College of Chemistry, Chemical Engineering and Biotechnology, Donghua University, Shanghai 201620, China -
Supported by:the National Natural Science Foundation of China, No. 51373031; the Program for New Century Excellent Talents in University, No. NCET-12-0828; the Science and Technology Commission of Shanghai Municipality, No. 15520720800; and the Fundamental Research Funds for the Central Universities
CLC Number:
Cite this article
Li Xue, Tang Jing-yu, Bao Lu-han, Chen Lin, Hong Feng.
share this article
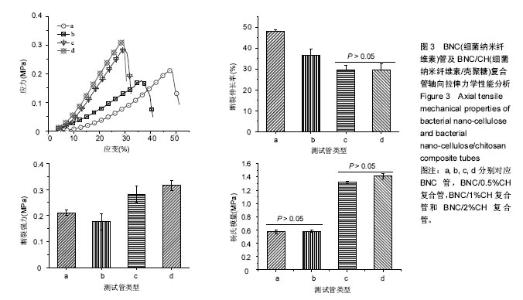
2.4 轴向拉伸力学测试结果 BNC管及BNC/CH复合管的轴向拉伸力学测试结果如图3所示。 由图3A可以看出,BNC管和BNC/CH管在轴向拉伸上具有一定的伸长率,并且当施加的力逐渐增大时,其形变也逐渐增大。 由图3B可知,BNC管的断裂伸长率为(48.0±0.1)%,复合壳聚糖后,BNC/CH管的断裂伸长率减小,BNC/0.5% CH管的断裂伸长率为(36.4±0.3)%,BNC/1%CH管和BNC/2%CH管的断裂伸长率之间差异无显著性意义,分别为(29.7±0.2)%和(29.5±0.3)%。 由图3C和3D可知,与BNC管相比,BNC/0.5%CH管的断裂强力和杨氏模量无明显变化,而BNC/1%CH管和 BNC/1%CH管的断裂强力和杨氏模量均增大,但是两者之间差异无显著性意义。综合来说,复合壳聚糖可以使轴向力学有一定的增强,但是延展性和弹性则会受到不利影响。"

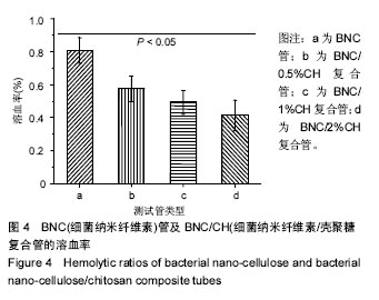
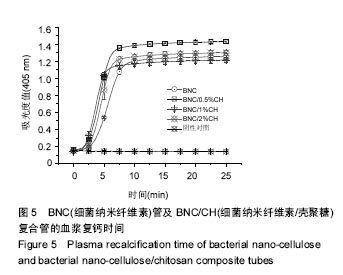
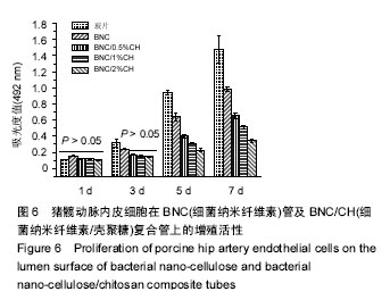
2.5 血液相容性 图4显示了BNC管及BNC/CH复合管对红细胞的影响,结果显示BNC管的溶血率为(0.8±0.1)%,壳聚糖沉积使管材的溶血率进一步减小,BNC/0.5%CH管、BNC/0.5%CH管和BNC/2%CH管的溶血率分别是(0.6±0.1)%、(0.5±0.1)%和(0.4±0.1)%,均低于1%。图5显示了BNC管及BNC/CH复合管对内源性凝血途径的激活程度,结果显示,BNC管对内源性凝血途径的激活最小,血浆复钙时间约为330 s,壳聚糖的沉积导致了对内源性凝血更大的激活,BNC/0.5%CH管、BNC/0.5%CH管和BNC/2%CH管的血浆复钙时间均显著减小,分别约为262 s、270 s和 275 s,但是不同复合管的血浆复钙时间并没有显著的不同。"

| [1]Kieser TM, Lewin AM, Graham MM, et al. Outcomes associated with bilateral internal thoracic artery grafting: the importance of age. Ann Thorac Surg. 2011;92(4):1269-1275.[2]Liu Y, Chen JR, Yang Y, et al. Improved blood compatibility of poly(ethylene terephthalate) films modified with L-arginine. J Biomater Sci Polym Ed. 2008;19(4):497-507.[3]Duan X, Lewis RS. Improved haemocompatibility of cysteine-modified polymers via endogenous nitric oxide. Biomaterials. 2002;23(4):1197-1203.[4]Stanley JC, Burkel WE, Ford JW, et al. Enhanced patency of small-diameter, externally supported Dacron iliofemoral grafts seeded with endothelial cells. Surgery. 1982;92(6):994-1005.[5]曹海建,钱坤.人造血管的现状及其研究进展[J]. 纺织科技进展, 2004(2): 31-33.[6]霍丹群,陈柄灿,侯长军,等.人工血管及其研究进展[J]. 中国医疗器械杂志,2004,28(3): 200-202.[7]Wu T, Jiang B, Wang Y, et al. Electrospun poly(L-lactide-co-caprolactone)/collagen/chitosan vascular graft in a canine femoral artery mode. Journal of Materials Chemistry B. 2015; 3(28): 5760-5768.[8]Huang C, Chen R, Ke Q, et al. Electrospun collagen-chitosan- TPU nanofibrous scaffolds for tissue engineered tubular grafts. Colloids Surf B Biointerfaces. 2011;82(2):307-315.[9]Cattaneo I, Figliuzzi M, Azzollini N, et al. In vivo regeneration of elastic lamina on fibroin biodegradable vascular scaffold. Int J Artif Organs.2013;36(3):166-174.[10]de Valence S, Tille JC, Giliberto JP, et al. Advantages of bilayered vascular grafts for surgical applicability and tissue regeneration. Acta Biomater. 2012;8(11):3914-3920.[11]谭玉静,洪枫,邵志宇. 细菌纤维素在生物医学材料中的应用[J]. 中国生物工程杂志, 2007,27(4):126-131.[12]洪枫. 用于急性创伤的细菌纤维素基抗菌干膜及其制备方法和应用: CN, ZL201110009676.3[P]. 中国发明专利, 2011.[13]Wei B, Yang G, Hong F. Preparation and evaluation of a kind of bacterial cellulose dry films with antibacterial properties. Carbohydrate Polymers. 2011; 84(1): 533-538.[14]Petersen N, Gatenholm P. Bacterial cellulose-based materials and medical devices: current state and perspectives. Appl Microbiol Biotechnol. 2011;91(5):1277-1286.[15]Gatenholm P, Klemm D. Bacterial nanocellulose as a renewable material for biomedical applications. Mrs Bulletin. 2010; 35(3):208-213.[16]洪枫,蒋燕光,陈琳,等. 小径纳米纤维素管的生物制备及其表征[J]. 纤维素科学与技术, 2014,22(3):1-7.[17]洪枫,魏斌,杨光. 一种制备中空异形细菌纤维素材料的装置: CN, ZL201020511755.5[P]. 中国实用新型专利, 2010.[18]Wippermann J, Schumann D, Klemm D, et al. Preliminary results of small arterial substitute performed with a new cylindrical biomaterial composed of bacterial cellulose. Eur J Vasc Endovasc Surg. 2009;37(5):592-596.[19]Bäckdahl H, Bo R, Gatenholm P. Observations on bacterial cellulose tube formation for application as vascular graft. Materials Science & Engineering C. 2011; 31(1): 14-21.[20]Putra A, Kakugo A, Furukawa H, et al. Tubular bacterial cellulose gel with oriented fibrils on the curved surface. Polymer. 2008; 49(7):1885-1891.[21]Azad AK, Sermsintham N, Chandrkrachang S, et al. Chitosan membrane as a wound-healing dressing: characterization and clinical application. J Biomed Mater Res B Appl Biomater. 2004;69(2):216-222.[22]Zhang P, Chen L, Zhang Q, et al. Using In situ Dynamic Cultures to Rapidly Biofabricate Fabric-Reinforced Composites of Chitosan/Bacterial Nanocellulose for Antibacterial Wound Dressings. Front Microbiol. 2016;7:260.[23]Uchegbu IF, Carlos M, Mckay C, et al. Chitosan amphiphiles provide new drug delivery opportunities. Polymer International. 2014; 63(7): 1145-1153.[24]Brewster LP, Bufallino D, Ucuzian A, et al. Growing a living blood vessel: insights for the second hundred years. Biomaterials. 2007;28(34):5028-5032.[25]Kong X, Han B, Li H, et al. New biodegradable small-diameter artificial vascular prosthesis: a feasibility study. J Biomed Mater Res A. 2012;100(6):1494-1504.[26]Kong X, Han B, Wang H, et al. Mechanical properties of biodegradable small-diameter chitosan artificial vascular prosthesis. J Biomed Mater Res A. 2012;100(8):1938-1945.[27]Tang J, Li X, Bao L, et al. Comparison of two types of bioreactors for synthesis of bacterial nanocellulose tubes as potential medical prostheses including artificial blood vessels. Journal of Chemical Technology & Biotechnology. 2017; 92: 1218-1228.[28]Leitão AF, Gupta S, Silva JP, et al. Hemocompatibility study of a bacterial cellulose/polyvinyl alcohol nanocomposite. Colloids Surf B Biointerfaces. 2013;111:493-502.[29]Wang S, Zhang Y, Yin G, et al. Electrospun polylactide/silk fibroin–gelatin composite tubular scaffolds for small-diameter tissue engineering blood vessels. Journal of Applied Polymer Science. 2010; 113(4): 2675-2682. |
| [1] | Yao Xiaoling, Peng Jiancheng, Xu Yuerong, Yang Zhidong, Zhang Shuncong. Variable-angle zero-notch anterior interbody fusion system in the treatment of cervical spondylotic myelopathy: 30-month follow-up [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(9): 1377-1382. |
| [2] | An Weizheng, He Xiao, Ren Shuai, Liu Jianyu. Potential of muscle-derived stem cells in peripheral nerve regeneration [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(7): 1130-1136. |
| [3] | Zhang Jinglin, Leng Min, Zhu Boheng, Wang Hong. Mechanism and application of stem cell-derived exosomes in promoting diabetic wound healing [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(7): 1113-1118. |
| [4] | Le Guoping, Zhang Ming, Xi Licheng, Luo Hanwen. Preparation and in vitro evaluation of vancomycin hydrochloride@polylactic acid-glycolic acid copolymer-chitosan-hyaluronic acid composite sustained-release microspheres [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(4): 528-534. |
| [5] | He Yunying, Li Lingjie, Zhang Shuqi, Li Yuzhou, Yang Sheng, Ji Ping. Method of constructing cell spheroids based on agarose and polyacrylic molds [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(4): 553-559. |
| [6] | He Guanyu, Xu Baoshan, Du Lilong, Zhang Tongxing, Huo Zhenxin, Shen Li. Biomimetic orientated microchannel annulus fibrosus scaffold constructed by silk fibroin [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(4): 560-566. |
| [7] | Chen Xiaoxu, Luo Yaxin, Bi Haoran, Yang Kun. Preparation and application of acellular scaffold in tissue engineering and regenerative medicine [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(4): 591-596. |
| [8] | Kang Kunlong, Wang Xintao. Research hotspot of biological scaffold materials promoting osteogenic differentiation of bone marrow mesenchymal stem cells [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(4): 597-603. |
| [9] | Shen Jiahua, Fu Yong. Application of graphene-based nanomaterials in stem cells [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(4): 604-609. |
| [10] | Zhang Tong, Cai Jinchi, Yuan Zhifa, Zhao Haiyan, Han Xingwen, Wang Wenji. Hyaluronic acid-based composite hydrogel in cartilage injury caused by osteoarthritis: application and mechanism [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(4): 617-625. |
| [11] | Li Hui, Chen Lianglong. Application and characteristics of bone graft materials in the treatment of spinal tuberculosis [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(4): 626-630. |
| [12] | Gao Cangjian, Yang Zhen, Liu Shuyun, Li Hao, Fu Liwei, Zhao Tianyuan, Chen Wei, Liao Zhiyao, Li Pinxue, Sui Xiang, Guo Quanyi. Electrospinning for rotator cuff repair [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(4): 637-642. |
| [13] | Guan Jian, Jia Yanfei, Zhang Baoxin , Zhao Guozhong. Application of 4D bioprinting in tissue engineering [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(3): 446-455. |
| [14] | Liu Jiali, Suo Hairui, Yang Han, Wang Ling, Xu Mingen. Influence of lay-down angles on mechanical properties of three-dimensional printed polycaprolactone scaffolds [J]. Chinese Journal of Tissue Engineering Research, 2022, 10(16): 2612-2617. |
| [15] | Huang Bo, Chen Mingxue, Peng Liqing, Luo Xujiang, Li Huo, Wang Hao, Tian Qinyu, Lu Xiaobo, Liu Shuyun, Guo Quanyi . Fabrication and biocompatibility of injectable gelatin-methacryloyl/cartilage-derived matrix particles composite hydrogel scaffold [J]. Chinese Journal of Tissue Engineering Research, 2022, 10(16): 2600-2606. |
| Viewed | ||||||
|
Full text |
|
|||||
|
Abstract |
|
|||||